US Duchenne Muscular Dystrophy (DMD) Therapeutics Market Analysis
US Duchenne Muscular Dystrophy (DMD) Therapeutics Market is projected to grow from $xx Mn in 2023 to $xx Mn by 2030, registering a CAGR of xx% during the forecast period of 2023 ? 2030. A genetic condition known as Duchenne muscular dystrophy (DMD) is characterised by advancing muscle weakness and degeneration as a result of changes to a protein called dystrophin, which aids in maintaining healthy muscle cells. The market is expanding as a result of favourable government initiatives that provide appropriate reimbursement policies and promote target-specific treatments. Rapid urbanisation, better medical infrastructure, the emergence of product premiumization, and rising disposable incomes among the general populace are a few other drivers generating profitable growth potential in the industry. Sarepta Therapeutics, PTC Therapeutics, Pfizer, Bristol-Myers Squibb, Italfarmaco, Santhera Pharmaceuticals, and many more are Global companies in the DMD Therapeutics market.
Buy Now

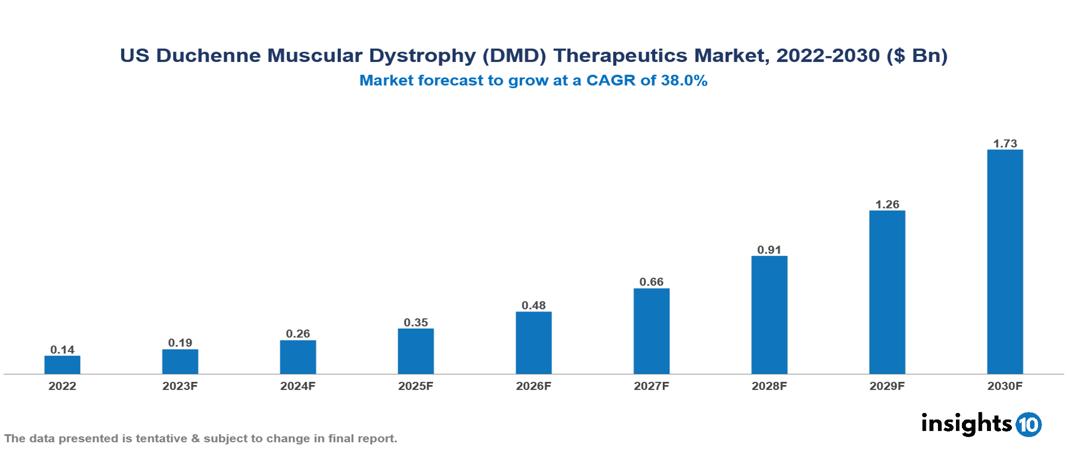
US Duchenne Muscular Dystrophy (DMD) Therapeutics Market Analysis Summary
US Duchenne Muscular Dystrophy (DMD) Therapeutics Market is valued at around $0.14 Bn in 2022 and is projected to reach $1.73 Bn by 2030, exhibiting a CAGR of 38% during the forecast period 2023-2030.
A genetic condition known as Duchenne muscular dystrophy (DMD) is characterised by advancing muscle weakness and degeneration as a result of changes to a protein called dystrophin, which aids in maintaining healthy muscle cells. One of the four diseases referred to as dystrophinopathies is DMD. The other three disorders in this group include DMD-associated dilated cardiomyopathy (heart disease) with little to no clinical skeletal, or voluntary, muscle dysfunction, Becker muscular dystrophy (BMD, a mild variant of DMD), and an intermediate clinical presentation between DMD and BMD. The main sign of DMD is muscle weakness. It can start as early as 2 or 3 years old, initially affecting the proximal muscles (those closest to the body's centre) before progressing to the distal limb muscles (those close to the extremities). The lower external muscles typically experience pain before the upper external muscles. The impacted child may have trouble running, walking, and leaping. Other signs include lumbar lordosis, swelling of the calves, and a waddling gait (an inward curve of the spine). Later, the respiratory and cardiac muscles are also impacted. DMD is inherited from the mother, who is known as a carrier, and has an X-linked recessive inheritance pattern.
Early childhood is when DMD symptoms first appear, usually between the ages of 2 and 3. Although it rarely happens, the condition primarily affects boys. The market is expanding as a result of favourable government initiatives that provide appropriate reimbursement policies and promote target-specific treatments. Rapid urbanisation, better medical infrastructure, the emergence of product premiumization, and rising disposable incomes among the general populace are a few other drivers generating profitable growth potential in the industry. Sarepta Therapeutics, PTC Therapeutics, Pfizer, Bristol-Myers Squibb, Italfarmaco, Santhera Pharmaceuticals, and many more are Global companies in the DMD Therapeutics market.
Market dynamics
Drivers of US Duchenne Muscular Dystrophy (DMD) Therapeutics Market
The market's expansion is significantly influenced by the increasing number of new products being approved and introduced by the key manufacturers. This is explained by the rising prevalence of Duchenne muscular dystrophy in the general population. In accordance with this, a significant increase in clinical trials and a robust product pipeline are energising the market.
Additionally, the introduction of mutation-specific medicines as a result of ongoing diagnostic innovation is favourably affecting the industry. In addition, the market is being driven by intensive research and development (R&D) initiatives focused on accurate DMD diagnosis and treatment for underrepresented populations, including newborns, women, and non-ambulant patients.
Development in Duchenne Muscular Dystrophy (DMD) Therapeutics Market
The FDA approved Viltepso in August 2020 for the treatment of people with a confirmed mutation of the DMD gene that is amenable to a therapeutic approach known as exon 53 skipping and may help up to 8% of people with DMD. Viltepso is an "exon skipping" drug that targets a section of DNA called exon 53.
For the treatment of Duchenne muscular dystrophy, DTDEC-01 is being created (DMD). Dystrophin-expressing chimeric (DEC) cells are the potential treatment. It is made up of ex-vivo fused autologous mesenchymal stem cells from human bone marrow and allogeneic myoblasts carrying the dystrophin gene. The treatment improves engraftment and does away with immunosuppression. Through the intraarticular (intra-bone) method, it is delivered.
Sarepta Therapeutics, a Cambridge, Massachusetts-based company, produces the treatment under the trade name SRP-9001. It would be the FDA's 13th approved gene therapy since 2017 and the first to focus on a common genetic illness in kids. Based on evidence that the therapy enables boys to produce an engineered form of dystrophin, the fast approval would allow the medicine to reach the market before significant clinical trials have been finished.
Emflaza was approved by the FDA on Friday for DMD patients as young as two years old. The medication had previously been authorised for use in patients older than five.
In a mouse model of Duchenne muscular dystrophy (DMD), researchers at the University of British Columbia (UBC) School of Biomedical Engineering discovered that inhibiting the colony-stimulating factor 1 receptor (CSF1R), a drug class already used in clinical trials to treat uncommon forms of cancer, increased muscle toughness and slowed DMD's progression.
Key players
Sarepta Therapeutics Solid Biosciences UniQure N.V. Pfizer Inc. Bristol-Myers Squibb PTC Therapeutics BioMarin Pharmaceutical Inc. Astellas Pharma Inc. Spark Therapeutics Orchard Therapeutics1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Market segmentations for US Duchenne Muscular Dystrophy (DMD) Therapeutics Market
By Product Type
- Corticosteroids
- Prednisolone
- Prednisone
- Deflazacort
- Pain Management Drugs
By Therapeutic Approach
- Mutation Suppression
- Exon Skipping
- Steroid Therapy
- Others
By End User
- Hospitals
- Clinics
- Home Care Settings
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.







































