Spain Mucopolysaccharidosis I (MPS-I) Drugs Market Analysis
Spain Mucopolysaccharidosis I (MPS-I) Drugs Market is projected to grow from $xx Mn in 2023 to $xx Mn by 2030, registering a CAGR of xx% during the forecast period of 2023 ? 2030. Rising consumer awareness, new drug approvals, enticing reimbursement plans, an established healthcare system, and the presence of sizable market competitors all drive the market for treating Mucopolysaccharidosis I. Major global players in Mucopolysaccharidosis I (MPS-I) Drugs Market are Takeda Pharmaceutical Company Limited, BioMarin Pharmaceuticals, Inc., Ultragenyx Pharmaceutical Inc., Sarepta Drugs, Abeona Drugs, Eloxx Pharmaceuticals, Esteve, Immusoft Corporation, Inventiva, GC Pharma, JCR Pharmaceuticals Co Ltd., RegenxBio Inc., Sangamo Drugs, Inc., Bioasis Technologies Inc., and Paradigm Biopharmaceuticals Ltd.
Buy Now

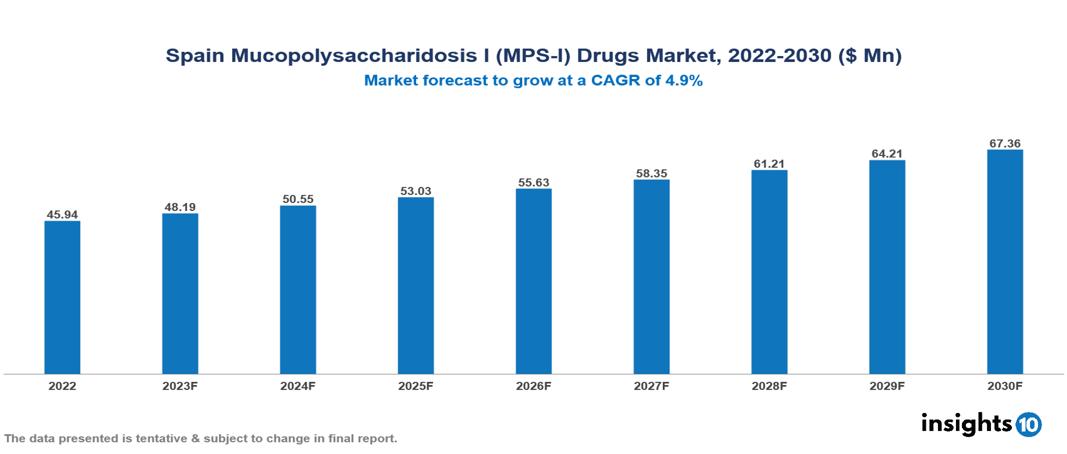
Spain Mucopolysaccharidosis I (MPS-I) Drugs Market Analysis Summary
Spain Mucopolysaccharidosis I (MPS-I) Drugs Market is valued at around $45.94 Mn in 2022 and is projected to reach $67.36 Mn by 2030, exhibiting a CAGR of 4.9% during the forecast period 2023-2030.
Alpha-L-iduronidase, a lysosomal enzyme ordinarily necessary for the breakdown of specific complex carbohydrates known as glycosaminoglycans (GAGs), is absent in MPS I (Mucopolysaccharidosis I), a hereditary lysosomal storage disorder. The spine's aberrant bones, a claw hand, clouded corneas, hearing loss, and heart valve issues are among the symptoms. Gene therapy, enzyme therapy, and bone marrow transplantation are all forms of treatment.
Mucopolysaccharides, a long chain of sugar molecules found throughout the body, are difficult for the body to effectively break down in a group of inherited diseases known as mucopolysaccharidosis. In consequence, these sugars accumulate in cells, blood, and connective tissues, which can lead to a number of health complications.
The treatment approach for MPS-I involves enzyme replacement therapy (ERT) and hematopoietic stem cell transplantation (HSCT). Aldurazyme (Laronidase) is an ERT which supplies the deficient enzyme alpha-L-iduronidase and by this severity of symptoms is decreased. HSCT (Hematopoietic Stem Cell Transplantation) in this patient's defective bone marrow is replaced with healthy donor cells that can produce the missing enzyme.
Major global players in Mucopolysaccharidosis I (MPS-I) Drugs Market are Takeda Pharmaceutical Company Limited, BioMarin Pharmaceuticals, Inc., Ultragenyx Pharmaceutical Inc., Sarepta Drugs, Abeona Drugs, Eloxx Pharmaceuticals, Esteve, Immusoft Corporation, Inventiva, GC Pharma, JCR Pharmaceuticals Co Ltd., RegenxBio Inc., Sangamo Drugs, Inc., Bioasis Technologies Inc., and Paradigm Biopharmaceuticals Ltd.
Market Dynamics
Market Drivers
The industry's top rivals are focusing on getting approval for their innovative treatments, which is expected to drive market growth over the course of the projected period. For example, in June 2019, the National Medical Products Administration (NMPA) approved Vimizim (elosulfase alfa) by Biomarine Pharmaceuticals Inc. for the treatment of patients with mucopolysaccharidosis type IVA (MPS IVA), also known as Morquio A syndrome. Vimizim is the name of the first treatment for this sickness in China.
Participants in the market for treating mucopolysaccharidosis (MPS) are putting more emphasis on implementing inorganic growth tactics, like partnerships and collaborations, in order to enhance their positions. Rising consumer awareness, enticing reimbursement plans, and an established healthcare system are all of these factors that act as market growth drivers.
Market Restraints
High therapy costs and insufficient diagnosis are expected to limit the growth of the global mucopolysaccharidosis (MPS) treatment market over the anticipated timeframe. One of the drugs used to treat MPS is venison (elosulfase alfa), whose annual cost was initially projected to be $380,000.
The rarity of the Disease, Complex Disease Management because MPS-I affects various organs, including the skeleton, heart, respiratory system, and central nervous system, Limited Treatment Options, Disease Progression and Diagnosis Challenges all these factors act as market growth restraints.
Key players
Vertex Pharmaceuticals AbbVie Genentech PTC Therapeutics Gilead Sciences AstraZeneca Boehringer Ingelheim Chiesi Farmaceutici Orphan Europe Catalyst Pharmaceuticals1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Market Segmentations For Spain Mucopolysaccharidosis I (MPS-I) Drugs Market
By Treatment :
- Enzyme Replacement Therapy
- Stem Cell Therapy
By Disease Type :
- MPS-I
- MPS-II
- MPS-IV
- MPS-VI
- MPS-VII
- Others (MPS-III and MPS-IX)
By End Users
- Hospitals
- Speciality Clinic
- Others
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.








































