Mexico Patient Adherence Programs Market Analysis
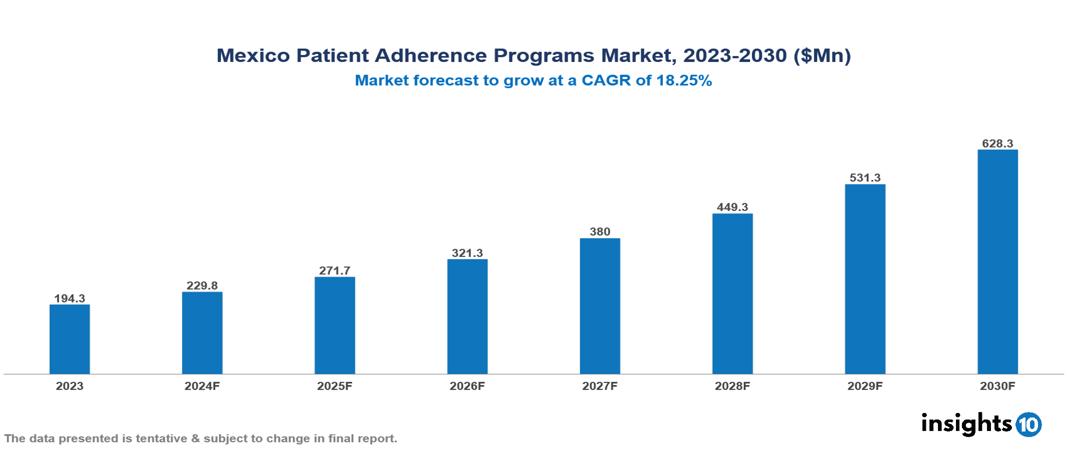
The Mexico Patient Adherence Programs Market was valued at $194.3 Mn in 2023 and is predicted to grow at a CAGR of 18.25% from 2023 to 2030, to $628.3 Mn by 2030. The key drivers of the market include. The prominent players in the Mexico Patient Adherence Programs Market are Grupo Neolpharma, PiSA Farmaceutica, Liomont, Laboratorios Sanfer, and Laboratorios Sophia, among others.
Buy Now

Mexico Patient Adherence Programs Market Executive Summary
The Mexico Patient Adherence Programs market is at around $194.3 Mn in 2023 and is projected to reach $628.3 Mn in 2030, exhibiting a CAGR of 18.25% during the forecast period.
A patient adherence program aims to ensure that patients follow prescribed medication regimens to improve treatment outcomes, using both direct and indirect methods to assess adherence. Direct methods include monitoring therapy by measuring drug levels, metabolites, or biological markers in blood or urine, and confirming medication intake. Indirect methods, more commonly used, involve patient self-reports, pill counts, prescription refill rates, clinical response evaluations, and electronic medication monitors. Pill counts compare the number of pills taken between appointments with the prescribed dosage, while patient self-reports collect information through interviews, questionnaires, or diaries. Electronic devices such as pill bottles or blister packs track medication access to provide precise data. A widely used tool for assessing adherence is the Morisky Medication Adherence Scale (MMAS), a validated and reliable questionnaire suitable for clinical use. These approaches help healthcare providers ensure consistent medication use, ultimately enhancing patient health outcomes.
The Mexico Patient Adherence Program Market is thus driven by significant factors such as increasing non-adherence, rising chronic conditions, and an aging population. However, inadequate healthcare infrastructure, high implementation costs, and limited accessibility restrict the growth and potential of the market.
The major players in the Mexico Patient Adherence Programs Market are Grupo Neolpharma, PiSA Farmaceutica, Liomont, Laboratorios Sanfer, and Laboratorios Sophia, among others.
Market Dynamics
Market Growth Drivers
Increasing Non-Adherence: The medical field is focusing more on developing workable solutions as a result of growing recognition of the substantial negative effects of non-adherence on patient outcomes. Governments and healthcare organizations’ awareness campaigns and actions highlight the value of adherence, which increases demand and growth for patient adherence programs.
Rising Chronic Conditions: Chronic diseases such as diabetes, hypertension, and heart disease require ongoing treatment and long-term medication adherence. The need for continuous monitoring and consistent medication use in chronic conditions necessitates adherence programs to ensure patients follow their prescribed regimens. Patient adherence programs lead to better clinical outcomes and enhanced quality of life, which creates a positive effect on the market growth.
Aging Population: Mexico’s population aged 65 and older is projected to grow by 277% from 8.2 Mn in 2015 to over 30 Mn by 2050. The elderly population is more prone to risk from chronic diseases and multiple comorbidities, necessitating complex medication regimens that are difficult to manage without structured adherence programs. Adherence programs help with polypharmacy and provide monitoring and support. The aging population is thus a potential pool that can benefit from these programs which increases the market growth.
Market Restraints
Inadequate Healthcare Infrastructure: By restricting access to vital healthcare services and resources required for these programs to be successful, inadequate healthcare infrastructure impedes the expansion of patient adherence initiatives. Patients face challenges in adhering to recommended therapies on a regular basis due to inadequate technology support, a lack of healthcare personnel, and poorly equipped facilities. Furthermore, inadequate infrastructure frequently results in healthcare practitioners' inability to adequately monitor and promote patient adherence, which has a negative impact on patient outcomes and diminishes confidence in these programs. As a result, these obstacles hinder market expansion by discouraging investments and limiting the efficacy and scalability of patient adherence programs.
High Implementation Costs: Establishing advanced technologies like electronic health records (EHRs), mobile health applications, telemedicine platforms, and electronic monitoring devices necessitates significant financial outlays. Additionally, the need to hire specialized personnel, such as IT experts, adherence counselors, and extra healthcare providers, adds to operational expenses. Integrating these new adherence technologies with existing healthcare systems and EHRs can be particularly complex and expensive. Thus, these all expenses related to patient adherence programs can negatively affect the market.
Limited Accessibility: Accessibility issues can hinder the expansion of the patient adherence program market, especially in regions with inadequate healthcare infrastructure. Patients in these areas often encounter obstacles like long travel distances to clinics, a scarcity of healthcare professionals, and limited access to affordable medications and adherence support. Socioeconomic challenges, such as low income and lack of insurance, further impede participation in adherence programs. These barriers reduce enrolment and engagement, thereby restricting the effectiveness and growth potential of these programs.
Regulatory Landscape and Reimbursement Scenario
In Mexico, the Federal Commission for Protection against Health Risks (COFEPRIS or Comisión Federal para la Protección contra Riesgos Sanitarios) is a regulatory body of the Mexican Government; a decentralized organ of the Mexican Secretariat of Health. Among the many health-related issues it regulates in Mexico are pharmaceuticals, food safety, medical devices, organ transplants, and environmental preservation. COFEPRIS is in charge of the entire drug approval process, from the initial application to the post-market surveillance (PMS) stage, including supply and demand, advertising, commercialization, imports and exports, distribution, manufacturing, and supply.
Mexico’s pharmaceutical industry is thriving and expanding, and thus strict laws are in place to guarantee the security and effectiveness of medications made available to Mexican residents. Pharmaceutical companies seeking to sell their medications in Mexico must understand the procedure thoroughly to get approval. The overall approval includes an extensive evaluation by COFEPRIS, accompanied by facility inspection to ensure GMP compliance. There are three different pathways for registering medicinal products with COFEPRIS. The New Drug Registration is the pathway applicable for new chemical entities that have not previously been marketed anywhere in the world. Next, is the Generic Drug Registration for drugs that are similar to the already-marketed reference drugs. Lastly, Biosimilar Registration is applicable to biological products that are similar to the approved reference biological product.
The healthcare reimbursement system in Mexico is characterized by a complex interplay between the public and private sectors. For the public sector, social security institutions are the main provider of public health insurance. Workers in the formal sector and their dependents are covered by the Instituto Mexicano del Seguro Social (IMSS). Government employees and their families are covered by the Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado (ISSSTE). To supplement public plans with extra coverage, many Mexicans opt to buy private health insurance which provides access to a greater variety of healthcare providers, such as private hospitals and specialists. The healthcare reimbursement scheme in Mexico is gradually evolving. Although many people have a safety net in public services, there are still large differences. In the future, there may be initiatives to increase public-private partnerships, achieve universal coverage, and use technology to improve healthcare affordability and accessibility for all.
Competitive Landscape
Key Players
Here are some of the major key players in the Mexico Patient Adherence Programs Market:
- Grupo Neolpharma
- PiSA Farmaceutica
- Liomont
- Laboratorios Sanfer
- Laboratorios Sophia
- Genomma Lab Internactional
- Laboratorios Silanes
- Profarma
- Landsteiner Scientific
- Carnot Laboratories
1. Executive Summary
1.1 Service Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Healthcare Services Market in Country
1.6 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Market Size (With Excel and Methodology)
2.2 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Services
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Mexico Patient Adherence Programs Market Segmentation
By Type
- Hardware centric
- Software centric
By Medication
- Cardiovascular
- Nervous System
- Diabetes
- Gastrointestinal
- Oncology
- Rheumatology
- Others
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.







































