Japan Rituximab Biosimilars Market Analysis
Japan Rituximab Biosimilars Market is projected to grow from $xx Mn in 2022 to $xx Mn by 2030, registering a CAGR of xx% during the forecast period of 2022 - 2030. Rituximab biosimilars market is growing globally due to the government initiatives, increased healthcare expenditure, increase in biopharmaceutical R&D expenditure, low cost of biosimilars, aging population, increase in healthcare access, patent expiry of reference product, rising prevalence of chronic diseases and growing acceptance and confidence for biosimilars. Teva Pharmaceuticals, Novartis AG, Pfizer, BIOCAD, and Shanghai Henlius Biotech, Inc. are the key global market players in Rituximab Biosimilars Market.
Buy Now

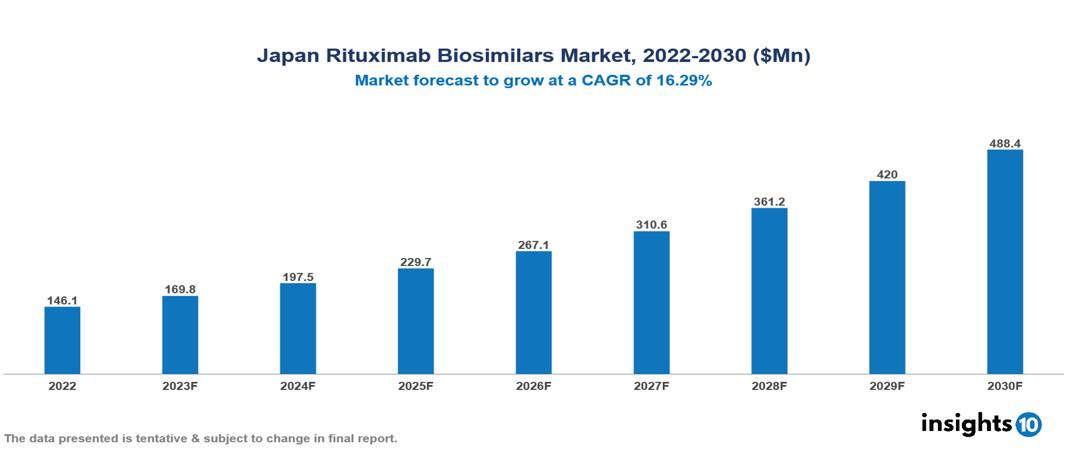
Japan Rituximab Biosimilars Market Analysis Summary
Japan Rituximab Biosimilars Market is valued at around $146.1 Mn in 2022 and is projected to reach $488.4 Mn by 2030, exhibiting a CAGR of 16.29% during the forecast period 2023-2030.
Rituximab is a chimeric anti-CD20 monoclonal antibody typically used to treat chronic lymphocytic leukemia, non-Hodgkin’s lymphoma, and rheumatoid arthritis. Rituxan is the trade name for rituximab. Rituximab is primarily administered by subcutaneous bolus injection or through an intravenous infusion. Rituximab biosimilars are biologic medications that closely resemble the reference product, Rituximab, a monoclonal antibody. To be comparable to the reference product in terms of quality, efficacy, and safety, biosimilars are produced. The EMA authorized CT-P10 (Truxima), the first biosimilar, in 2017. As a reference biological medicine, this medication has been approved for the treatment of all conditions. The EMA authorized GP2013 (Rixathon), a second biosimilar, for the treatment of rheumatoid arthritis. Ruxience, the third licensed rituximab biosimilar, is largely employed to treat lymphoma and leukemia. Teva Pharmaceuticals, Novartis AG, Pfizer, BIOCAD, and Shanghai Henlius Biotech, Inc. are the key global market players in Rituximab Biosimilars Market.
Market Dynamics
Market Growth Drivers
Increased healthcare expenditure, increase in biopharmaceutical R&D expenditure, low cost of biosimilars, aging population, increase in healthcare access, patent expiry of the reference product, rising prevalence of chronic diseases, and growing acceptance and confidence for biosimilars. All these factors act as market growth drivers.
Market Restraints
Fewer drug approvals, stringent government regulations, and lack of awareness are the suspected reasons for hindering the growth of the Rituximab biosimilar market.
Competitive Landscape
Key Players
- Teva Pharmaceuticals
- Novartis AG
- Pfizer
- BIOCAD
- Shanghai Henlius Biotech, Inc.
1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Market Segmentations For Japan Rituximab Biosimilars Market
By Product type
- Blitzima
- Rixathon
By Indication
- Non-Hodgkin’s Lymphoma
- Chronic lymphocytic leukaemia
- Rheumatoid arthritis
By distribution channel
- Hospital-based pharmacies
- Retail pharmacies
- Online pharmacies
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.








































