Indonesia Immune Checkpoint Inhibitors Market Analysis
Indonesia immune checkpoint inhibitors market is projected to grow from $xx Mn in 2022 to $xx Mn by 2030, registering a CAGR of xx% during the forecast period of 2022 - 2030. The market for immune checkpoint inhibitors is expanding as a result of factors such as leading an unhealthy lifestyle, a rise in cancer incidence, and genetic mutation and alteration that have an impact on how much the nation's healthcare systems cost. Some of the global key players in this market are AstraZeneca PLC, Eli Lilly and Company (ARMO Biosciences.), Bristol-Myers Squibb Company, Roche Holding AG, Incyte Corporation, Novartis AG, etc.
Buy Now

Indonesia Immune Checkpoint Inhibitors Market Analysis Summary
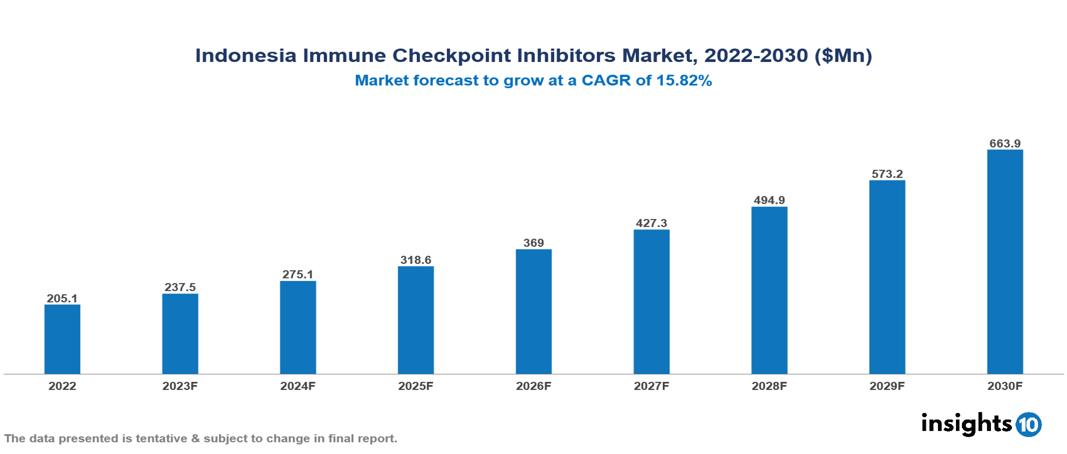
Indonesia Immune Checkpoint Inhibitors Market is valued at around $205.1 Mn in 2022 and is projected to reach $663.9 Mn by 2030, exhibiting a CAGR of 15.82% during the forecast period 2023-2030.
Some tumours are treated with radiation and surgery, and oncology is a big field with many of different applications. The bloodstream is routinely used to deliver chemotherapy systemically, or all across the patient's body. After receiving treatment for malignant cells, patients' immune systems fight cancer more successfully. The top cause of death in the world is cancer. Immune checkpoint inhibitors do this by inhibiting the growth of cancer cells and assisting the immune system or cells in identifying them. Drugs that suppress immune checkpoints specifically target immunological cells that must be turned on and off in order to initiate an immune response and prevent immune system damage. The MSI-H and dMMR malignancies are treated with the checkpoint inhibitor pembrolizumab, which was the first drug approved by the FDA solely based on the presence of a genetic characteristic in a tumour. Immune checkpoint inhibitors are seeing growth in the market as a result of escalating industrial competitiveness. The market for immune checkpoint inhibitors is expanding as a result of factors such as leading an unhealthy lifestyle, a rise in cancer incidence, and genetic mutation and alteration that have an impact on how much the nation's healthcare systems cost. The global key players in this market are AstraZeneca PLC, Eli Lilly and Company (ARMO Biosciences.), Bristol-Myers Squibb Company, Roche Holding AG, Incyte Corporation, Novartis AG, GlaxoSmithKline PLC, F. Hoffmann-La Roche Ltd. (Genentech Inc.,), Sanofi, Merck & Co., Inc., Merck KGaA (EMD Serono Inc.), BeiGene Ltd, Shanghai Jhunsi Biosciences Ltd.
Market Dynamics
Market Growth Drivers
Immune checkpoint inhibitors are experiencing growth in the market as a result of heightened industrial competitiveness. The market for immune checkpoint inhibitors is expanding as a result of factors that influence the expense of the nation's healthcare systems, such as an unhealthy lifestyle, an increase in cancer incidence, and genetic mutation and change. Rising healthcare expenses, increased use of immune checkpoint inhibitor medications in developing nations, and technological developments in cancer screening and dissemination methods are all contributing to the growth of the immune checkpoint inhibitor market.
Market Restraints
Regulatory price controls have also been brought about by the expensive nature of immunological checkpoint medicines. Another aspect that can limit market expansion is the immune checkpoint inhibitors' regulatory price ceiling. The market for immune checkpoint inhibitors is being constrained by both an increase in public awareness of immune system disorders and technological advancements in cancer therapy.
Competitive Landscape
Key Players
- AstraZeneca PLC
- Eli Lilly and Company (ARMO Biosciences.)
- Bristol-Myers Squibb Company
- Roche Holding AG
- Incyte Corporation
- Novartis AG
- GlaxoSmithKline PLC
- F. Hoffmann-La Roche Ltd. (Genentech Inc.,)
- Sanofi
- Merck & Co., Inc.
- Merck KGaA (EMD Serono Inc.)
- BeiGene Ltd.
- Shanghai Jhunsi Biosciences Ltd.
1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Indonesia Immune Checkpoint Inhibitors Market Segmentation
By Indications
- Breast cancer
- Bladder cancer
- Cervical cancer
- Hodgkin lymphoma
- Liver cancer
- Lung cancer
- Kidney cancer
- Others
By Target
- PD-1
- PD-L1
- CTLA-4
- Others
By Route of Administration
- Oral
- Parenterals
- Others
By End Users
- Hospitals
- Specialty clinics
- others
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.






































