China Cardiac Surgery Instruments Market Analysis
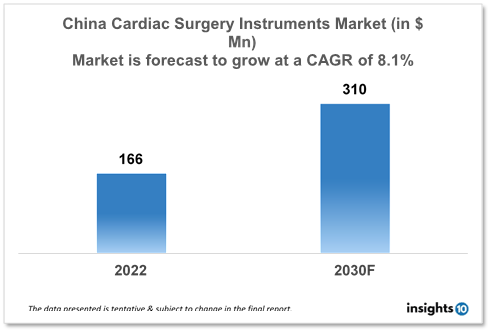
The China Cardiac Surgery Instruments Market is expected to witness growth from $166 Mn in 2022 to $310 Mn in 2030 with a CAGR of 8.10% for the forecasted year 2022-2030. In China, due to rising disposable income, more people in China can now access better healthcare services like cardiac surgeries. This has increased demand for cardiac surgery equipment, providing manufacturers with new business opportunities. The market is segmented by type, application and by end user. Some key players in this market include Lepu Medical Technology, MicroPort, LivaNova, B. Braun, Medline Industries, KLS Martin, and STILLE.
Buy Now

China Cardiac Surgery Instruments Healthcare Market Executive Analysis
The China Cardiac Surgery Instruments Market size is at around $166 Mn in 2022 and is projected to reach $310 Mn in 2030, exhibiting a CAGR of 8.10% during the forecast period. China had spent $46.05 billion on healthcare and family planning as of December 2022. This suggests an increase compared to previous forecasts of $28.02 Bn for November 2022. Prior to reaching a peak of $46.05 billion in December 2022, the figures reached a new low of $0.86 billion in January 2007. As a percentage of GDP in 2022, China's health spending was 5.4%. As a percentage of GDP, China's health spending grew from 4.9% of GDP in 2012 to 5.4% of GDP in 2022, growing at an average annual rate of 1.00%.
China has a mortality rate of around 30% due to Cardiovascular Diseases (CVDs). Ischemic heart disease, also known as coronary artery disease, is the most common type of CVD in China. About 40% of all CVD-related deaths in China are attributable to IHD. Dyslipidemia, which is defined by abnormally high levels of cholesterol and other lipids in the blood, affects about 40% of the population in China. Cardiovascular surgeons work on the heart and blood vessels using specialized equipment referred to as cardiac surgery instruments. These instruments are crucial for helping surgeons effectively complete a range of cardiac surgeries. The common cardiac procedure known as coronary artery bypass grafting (CABG) is used to boost the blood flow through the heart. Using instruments used in heart surgery, such as scissors, forceps, and needle holders, bypass grafts are joined to the coronary arteries during this procedure. A mechanical or biological valve is used during a valve replacement procedure to repair a diseased or damaged heart valve. During this procedure, cardiac surgery instruments like valve retractors, dilators, and sutures are used to remove the old valve and implant the new one.
Market Dynamics
Market Growth Drivers
In China, due to rising disposable income, more people in China can now access better healthcare services like cardiac surgeries. This has increased demand for cardiac surgery equipment, providing makers with new business opportunities. The proportion of people over 65 is increasing in China as the population ages. This group has a larger need for cardiac surgery equipment because they are at a higher risk of developing cardiovascular diseases. Market expansion is anticipated for makers who can appeal to this demographic. Technology advancements have led to the development of more advanced and cutting-edge cardiac surgery tools. Surgery is now more efficient as a result, which leads to quicker recovery periods and better patient outcomes. The manufacturers market presumably expand as a result of China healthcare market.
Market Restraints
In China, instruments for cardiac surgery require specialized materials and technology, which makes them costly to create and produce in China. Because of this, competing with larger companies that have more resources and access to economies of scale may be difficult for producers. Smaller manufacturers may find it difficult to innovate and create new goods because they have less access to funding for research and development. This may hamper their ability to develop their companies and compete in the China healthcare market for cardiac surgery instruments.
Competitive Landscape
Key Players
- Lepu Medical Technology (CN)
- MicroPort (CN)
- LivaNova
- B. Braun
- KLS Martin
- Medline Industries
- STILLE
Healthcare Policies and Regulatory Landscape
In China, a new regulatory structure for cardiac surgery instruments was put in place by China's National Medical Products Administration (NMPA). Stricter guidelines for clinical testing and registration of cardiac surgery instruments are included in this structure. Before releasing their cardiac surgery instruments on the Chinese market, companies must receive regulatory clearance. Recently, China has worked to strengthen its intellectual property protection regulations, which is significant for businesses selling cardiac surgery instruments. However, there is still a problem with intellectual property theft in China, so businesses must take precautions to safeguard their patents and brands. Good Manufacturing Practices (GMP) guidelines must be followed by cardiac surgery instrument manufacturers in China when creating and distributing their goods, according to regulations. This entails making sure that production facilities are spotless, that tools and instruments are packed and labelled correctly, and that quality control procedures are in place.
1. Executive Summary
1.1 Device Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Regulatory Landscape for Medical Device
1.6 Health Insurance Coverage in Country
1.7 Type of Medical Device
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Market Size (With Excel and Methodology)
2.2 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Cardiac Surgery Instruments Market Segmentation
By Type (Revenue, USD Billion):
The market is divided into segments in this study based on the goods, applications, end users, and geographical areas. The market is divided into forceps, scissors, needle holders, clamps, and other cardiac surgery instruments based on the product. In 2017 the forceps category led the market, and it is anticipated that it will increase at the fastest rate going forward. The rise in heart surgeries and the frequent usage of forceps in most cardiac procedures are credited with the segment's strong growth.
- Forceps
- Vascular Forceps
- Grasping Forceps
- Other Forceps
- Needle Holders
- Scissors
- Clamps
- Other Cardiac Surgical Instruments
By Application (Revenue, USD Billion):
The market is further segmented by application into paediatric cardiac surgery, heart valve surgery, coronary artery bypass graft (CABG), and other applications. The China market's largest and fastest-growing application segment is CABG. This is mostly explained by the increased prevalence of heart illnesses and the consequent rise in surgical treatments. The second-largest category is heart valve surgery.
- Coronary Artery Bypass Graft (CABG)
- Heart Valve Surgery
- Pediatric Cardiac Surgery
- Other Applications
By End User (Revenue, USD Billion):
Based on the end user, the market is segmented into hospitals and cardiac centers, and ambulatory surgery centers. The hospitals and cardiac centers segment is expected to dominate the market for cardiac surgery instruments. Growth in this end-user segment can be attributed to the increasing incidence of cardiac and heart valve diseases and the subsequent increase in the number of cardiac surgery procedures.
- Hospitals and Cardiac Centers
- Ambulatory Surgery Centers
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.









































