Canada Hunter Syndrome Therapeutics Market Analysis
Canada Hunter Syndrome Therapeutics Market is projected to grow from $xx Mn in 2023 to $xx Mn by 2030, registering a CAGR of xx% during the forecast period of 2023 - 2030. Rare X-linked recessive genetic disorder MPS II (Hunter Syndrome) is brought on by changes in the iduronate-2-sulfatase gene (IDS). Due to the poor understanding of the illness, creating effective treatments for uncommon diseases can be difficult and expensive, which has a negative impact on the market for syndromes. Global Key Players in the Hunter Syndrome Therapeutics Market Include Takeda Pharmaceutical Company Limited, BioMarin Pharmaceutical Inc., Shire (now a part of Takeda), Sanofi Genzyme, Ultragenyx Pharmaceutical Inc., Sangamo Therapeutics, Inc., RegenxBio Inc., Denali Therapeutics Inc., JCR Pharmaceuticals Co., Ltd., and ArmaGen Inc.
Buy Now

Canada Hunter Syndrome Therapeutics Market Analysis Summary
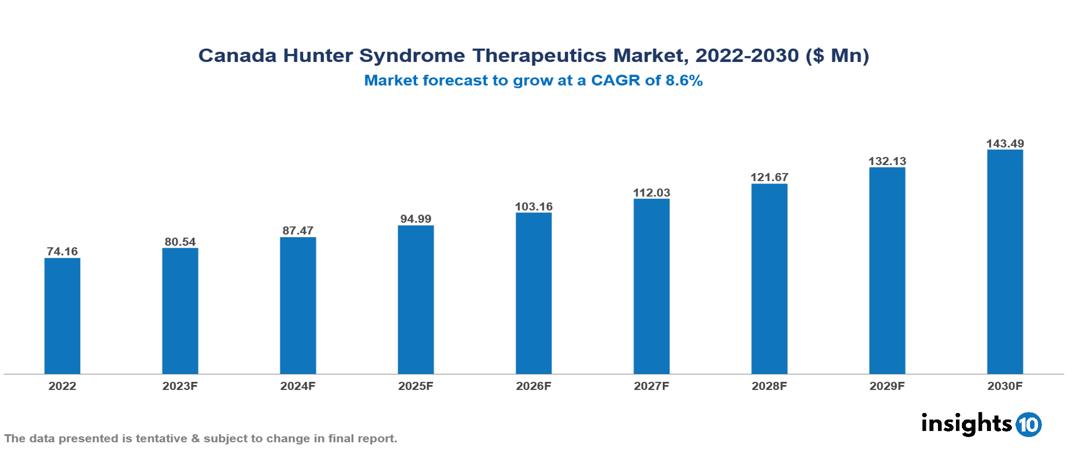
Canada Hunter Syndrome Therapeutics Market is valued at around $74.16 Mn in 2022 and is projected to reach $143.49 Mn by 2030, exhibiting a CAGR of 8.6% during the forecast period 2023-2030.
One of the set of illnesses known as mucopolysaccharidoses is Hunter syndrome. It is also referred to as MPS II, or mucopolysaccharidosis type II. A rare genetic condition called Hunter syndrome causes the body to improperly digest (break down) sugar molecules. These chemicals can harm physical and mental growth and capacities if they accumulate in organs and tissues over time. Boys virtually always experience the condition. Male patients are generally always the ones with Hunter syndrome. Approximately 1 out of 100,000 to 170,000 guys are diagnosed with it by doctors. It is possible for females to carry the genetic mutation that causes MPS II.
Due to the high cost of care, difficulties in making an accurate diagnosis, and the difficulty in creating viable treatments for uncommon diseases, the market for syndromes is constrained.
Global Key Players in the Hunter Syndrome Therapeutics Market Include BioMarin Pharmaceutical Inc., Shire (now a subsidiary of Takeda), Sanofi Genzyme, Ultragenyx Pharmaceutical Inc., Sangamo Therapeutics, Inc., RegenxBio Inc., Denali Therapeutics Inc., JCR Pharmaceuticals Co., Ltd., and ArmaGen Inc.
Market Dynamics
Market Restraints
High Treatment Cost: Because medicines for rare disorders like Hunter syndrome require extensive research and development, they can be expensive to buy and administer. For patients and healthcare systems, the high cost of treatment may result in accessibility difficulties, problems with reimbursement, and issues with affordability.
Challenges in Diagnosis: Hunter syndrome is a complex condition that may require specialized diagnostic tools and expertise. The challenges in accurately diagnosing the disease, particularly in its early stages, can result in delayed or missed diagnoses, hindering the timely initiation of treatment.
Research and Development Challenges: Developing effective treatments for rare diseases like Hunter syndrome can be challenging due to the limited understanding of the condition and the complex mechanisms involved. The rarity of the disease also poses challenges in conducting clinical trials with a sufficient number of participants.
Market Development
Genetic HMI-203 from Homology Medicine is undergoing a Phase I clinical trial. Intravenous administration is the method used to provide it.
Elaprase from Shire is undergoing a Phase IV clinical trial in people with Hunter syndrome, both adults and children (Mucopolysaccharidosis II).
Undergoing clinical trial research is Denali Therapeutics' DNL310, an investigational enzyme replacement therapy (ERT) that penetrates the central nervous system (CNS) and is intended to treat both the peripheral and CNS aspects of Mucopolysaccharidosis type II (MPS II; Hunter syndrome).
The only approved treatment for Hunter syndrome is enzyme replacement therapy (ERT) with recombinant idursulfase (ELAPRASE®); however, ERT as it is currently administered cannot cross the blood-brain barrier and is unable to meet the needs of MPS II patients with central nervous system (CNS) (neurocognition and behaviour) involvement. The goal of RGX-121 is to introduce a functioning gene into CNS cells. Pediatric patients are now participating in the clinical trial.
Key players
UniQure NV Pfizer Inc Spark Therapeutics Inc Orchard Therapeutics plc Bluebird Bio Inc Sangamo Therapeutics Inc Fate Therapeutics Inc Krystal Biotech Inc ProQR Therapeutics NV CRISPR Therapeutics AG1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Market Segmentations For Canada Hunter Syndrome Therapeutics Market
By Treatment
- Enzyme replacement drugs
- Stem cell transplant
- Therapy
- Physical therapy
- Speech therapy
By Diagnosis
- Urine test
- Blood test
- Genetic testing
By Distribution Channel
- Hospital
- Clinic
- Pharmacy
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.








































