Canada Biomaterials in Healthcare Market Analysis
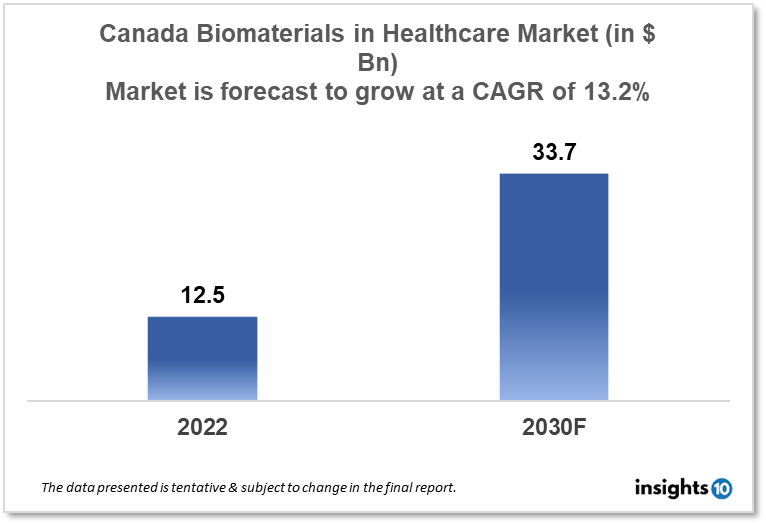
Canada's biomaterials in the healthcare market are likely to grow at a CAGR of 13.20% from a market size of $12.5 Bn in 2022 to $33.7 Bn in 2030. Market growth is driven mainly by factors such as increasing demand for medical implants and the sudden rise in cardiovascular diseases. This report is segmented by type and by application. Some key players in this market include Celanese Corporation, Berkeley Advanced Biomaterials, Stryker Corporation, Zimmer Biomet Holdings, and others.
Buy Now

Canada Biomaterials in Healthcare Market Executive Summary
Canada's Biomaterials in Healthcare Market size is at around $12.5 Bn in 2022 and is projected to reach $33.7 Bn in 2030, exhibiting a CAGR of 13.20% during the forecast period 2022-30. In 2021, Canada's total health spending amounted to $308 billion, or $8,019 per person.
A wide range of materials used in medical devices, drug delivery systems, tissue engineering, and regenerative medicine are included in the fast-expanding Canadian biomaterials in the healthcare market. Biomaterials are substances created to interact with biological systems, either to replace or improve a tissue or organ's functionality or to help carry medications and treatments to specific regions. The desire for cutting-edge and efficient medical devices and treatments in the Canadian healthcare market is what has fuelled the market's expansion for biomaterials. Metals, ceramics, polymers, and composites are just a few of the important biomaterials used in healthcare. The biomaterials market is divided into segments based on the applications they are used for, including orthopedics, cardiovascular, wound care, dental, plastic surgery, and others. The market for biomaterials is dominated by orthopedics due to the strong demand for spinal implants and joint replacements. The second-largest segment is cardiovascular, which is fuelled by the use of biomaterials in pacemakers, heart valves, and stents.
Market Dynamics
Market Growth Drivers
A number of factors are causing Canada's biomaterials business in healthcare to expand. Several of the main market factors for Canada's biomaterials sector in the healthcare sector are listed below:
Aging Population: The need for healthcare services, technologies, and biomaterials is being driven by Canada's aging population. People are more likely to require biomaterial-based medical implants and equipment as they get older. As a result, the market for Canadian producers of biomaterials is expanding.
Technical developments: New avenues for the creation of implants and medical devices have been made possible by advances in biomaterials technology. Manufacturers are now able to produce more robust, biocompatible, and useful biomaterials that may be employed in a range of applications thanks to new materials and fabrication methods.
Assistance from the government: The Canadian government has been aggressively promoting the growth of the nation's biomaterials sector in the healthcare sector. A supportive economic climate for Canadian manufacturers of biomaterials has been made possible through funding for research and development, tax breaks, and other support initiatives.
Growing Concerns About the Environmental Impact of Healthcare: As awareness of sustainability increases, so does the demand for sustainable biomaterials. With several businesses specializing in the creation of bio-based and environmentally friendly biomaterials, Canada's biomaterials industry is well-positioned to meet this need.
Market Restraints
Extensive research and development are often necessary for the creation of new biomaterials, which can be an expensive and time-consuming procedure. Smaller businesses may find it difficult to enter the market and compete with more established, larger competitors as a result. Even though the Canadian government has contributed some financing and support to the biomaterials sector, many businesses still struggle to find funding. Smaller businesses may find it more difficult to invest in R&D or grow as a result of this. When it comes to medical implants and gadgets, the healthcare sector can be slow to adopt new technologies. Even if they have created cutting-edge new materials or technologies, this can make it challenging for manufacturers of biomaterials to establish a presence in the market.
Competitive Landscape
Key Players
- Covalon Technologies
- Celanese Corporation
- Berkeley Advanced Biomaterials
- Stryker Corporation
- Zimmer Biomet Holdings
- Noble Biomaterials
- Evonik Industries
Healthcare Policies and Regulatory Landscape
Strict regulatory control is applied to the use of biomaterials in healthcare in Canada. The safety, effectiveness, and quality of medical devices and biomaterials used in healthcare are governed by the Canadian government through Health Canada. The use of biomaterials in medical devices and implants is governed by a number of laws and policies that Health Canada has put in place. They include the Medical Devices Regulations (MDR), which spells out the standards for medical devices' efficacy and safety in Canada. Provisions for biomaterials used in medical devices are included in the MDR, along with specifications for biocompatibility and other safety factors.
Health Canada has produced policies and guideline documents that offer direction to producers and users of biomaterials in addition to the MDR. One of them defines the risk classification system for medical devices and biomaterials in the Guideline Document for the Risk-based Classification System for Non-In Vitro Diagnostic Medical Devices (Non-IVDDs). Moreover, Health Canada offers guidelines for the assessment and testing of biomaterials for use in medical devices. This provides recommendations for conducting biocompatibility tests, which are used to assess the safety of biomaterials for use in implants and medical devices. In response to increasing dangers and developments in technology, the regulatory environment for biomaterials in healthcare is continually changing. New policies and regulations are being produced. In order to maintain compliance and the secure use of biomaterials, it is crucial for makers and users to stay current on Health Canada's most recent rules and guidance guidelines.
1. Executive Summary
1.1 Device Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Regulatory Landscape for Medical Device
1.6 Health Insurance Coverage in Country
1.7 Type of Medical Device
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Market Size (With Excel and Methodology)
2.2 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Canada Biomaterials in Healthcare Market Segmentation
By Type (Revenue, USD Billion):
Based on type, the market is segmented into Metallic Biomaterials, Polymeric Biomaterials, Ceramic Biomaterials, and Natural Biomaterials. The Metallic Biomaterials segment accounted for the largest share of the Canada market in 2019. The growing geriatric population Canadaly is expected to drive growth for this segment.
- Metallic Biomaterials
- Stainless Steel
- Titanium & Titanium Alloys
- Cobalt-Chrome Alloys
- Gold
- Silver
- Magnesium
- Polymeric Biomaterials
- Polymethylmethacrylate
- Polyethylene
- Polyester
- Silicone Rubber
- Nylon
- Polyetheretherketone
- Other Polymeric Biomaterials
- Ceramics
- Calcium Phosphate
- Zirconia
- Aluminum Oxide
- Calcium Sulfate
- Carbon
- Glass
- Natural Biomaterials
- Hyaluronic Acid
- Collagen
- Gelatin
- Fibrin
- Cellulose
- Chitin
- Alginates
- Silk
By Application (Revenue, USD Billion):
The cardiovascular, orthopaedic, dental, plastic surgery, wound healing, tissue engineering, ophthalmology, neurological/CNS, and other applications segments are made up of the biomaterials market. The market category for wound healing is anticipated to have the highest CAGR in 2019. The market will increase as a result of factors including expanding healthcare infrastructure, a large population pool, a rising diabetic population, and rising healthcare spending. Surgical Guides
- Cardiovascular
- Catheters
- Stents
- Implantable Cardiac Defibrillators
- Pacemakers
- Sensors
- Heart Valves
- Vascular Grafts
- Guidewires
- Others
- Orthopedic
- Joint Replacement
- Knee Replacement
- Hip Replacement
- Shoulder Replacement
- Others
- Viscosupplementation
- Bioresorbable Tissue Fixation
- Spine
- Spinal Fusion Surgeries
- Minimally Invasive Fusion Surgeries
- Motion Preservation & Dynamic Stabilization Surgeries
- Pedicle-Based Rod Systems
- Interspinous Spacers
- Artificial Discs
- Fracture Fixation Devices
- Bone Plates
- Screws
- Pins
- Rods
- Wires
- Synthetics Bone Grafts
- Joint Replacement
- Ophthalmology
- Contact Lenses
- Intraocular Lenses
- Functional Replacement of Ocular Tissues
- Synthetic Corneas
- Others
- Contact Lenses
- Dental
- Dental Implants
- Dental Bone Grafts & Substitutes
- Dental Membranes
- Tissue Regeneration
- Plastic Surgery
- Soft-Tissue Fillers
- Craniofacial Surgery
- Wound Healing
- Wound Closure Devices
- Sutures
- Staples
- Surgical Hemostats
- Internal Tissue Sealants
- Adhesion Barriers
- Hernia Meshes
- Wound Closure Devices
- Tissue Engineering
- Scaffolds for Regenerative Medicine
- Nanomaterials for Biosensing
- Tailoring of Inorganic Nanoparticles
- Neurological/Central Nervous System Applications
- Shunting Systems
- Cortical Neural Prosthetics
- Hydrogel Scaffolds for CNS Repair
- Neural Stem Cell Encapsulation
- Other Applications
- Drug Delivery Systems
- Gastrointestinal Applications
- Bariatric Surgery
- Urinary Applications
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.









































