Canada Allergy Therapeutics Market Analysis
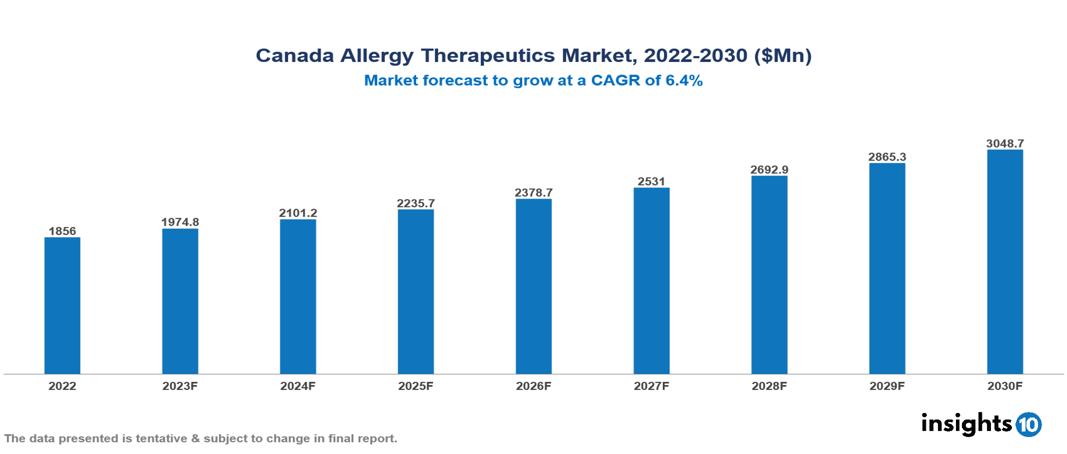
Canada Allergy Therapeutics Market was valued at $1856 Mn in 2022 and is estimated to reach $3049 Mn in 2030, exhibiting a CAGR of 6.4% during the forecast period. The rising incidence of allergic diseases and associated illnesses is a major market driver for the expansion of the allergy therapeutics market. Some of the leading pharmaceutical companies operating in the market are Sanofi, Novartis, GlaxoSmithKline (GSK), AstraZeneca, Merck & Co., Johnson & Johnson, Roche, Teva Pharmaceutical Industries, ALK-Abelló, and Stallergenes Greer
Buy Now

Canada Allergy Therapeutics Market Executive Summary
Canada Allergy Therapeutics Market was valued at $1856 Mn in 2022 and is estimated to reach $3049 Mn in 2030, exhibiting a CAGR of 6.4% during the forecast period.
Allergies are immune system sensitivities that arise from the body's reaction to environmental chemicals known as allergens, which are generally harmless to humans. In addition to food allergies like milk, eggs, soy, and almonds, these allergens can also be aeroallergens like dust mites, pollen, and mold. A runny nose and itchy eyes are examples of moderate allergy symptoms. Severe and potentially fatal allergy symptoms include anaphylaxis, which can result in unconsciousness and difficulty breathing. Immunotherapy, avoiding allergens, and prescription drugs, including nasal corticosteroids and antihistamines, are some of the treatments for allergies.
Food allergies are a major health concern in Canada, where 7% of people had food allergies, compared to 3-4% for adults and 5-6% for young children in westernized nations who have a physician's diagnosis. In Canada, 7–10% of children and 3–4% of adults suffer from allergies. The most common allergens are peanuts, tree nuts, milk, eggs, shellfish, soy, wheat, and sesame. Along with contact dermatitis, hives, and medication allergies, eczema is the most common skin allergy, affecting 10-15% of children and 3-5% of adults. Notable respiratory allergies include seasonal allergies to mold and pollen, which affect 15-20% of Canadians, asthma, which affects 8% of the population; and allergic rhinitis, or hay fever. Latex allergies are increasing, particularly among healthcare workers, and insect sting allergies, though less common, can be severe.
Sanofi, a renowned pharmaceutical giant, is actively working to advance allergy medicines, especially for children and adolescents with peanut allergies. To assess the safety and effectiveness of dupilumab, also known as Dupixent, Sanofi is presently carrying out a Phase 3 clinical investigation. The primary objective of this experiment is to evaluate dupilumab's potential as a peanut allergy therapy for the targeted age group.
Market Dynamics
Market Growth Drivers
Growing Prevalence of Allergies: The demand for allergy treatments has surged due to the rising number of patients suffering from a variety of allergies, including rhinitis, asthma, skin allergies, and allergies of the eyes. Pharmaceutical firms that specialize in allergy treatments are expecting an increase in demand for their goods and services as the number of people with allergies rises, which will propel the market's expansion.
Technological and Therapeutic Advancements: The creation of cutting-edge treatments such as monoclonal antibodies like dupilumab, sublingual immunotherapy (SLIT), and allergen-specific immunotherapy (AIT) offers more individualized and efficient treatment choices, attracting patients and growing the market.
Government Support and Initiatives: Supportive conditions for market expansion are produced by government by funding for allergy research and public health initiatives like the CIHR-Allergy Canada Research Network that target allergy prevention and management.
Market Restraints
High Cost of Treatment: A substantial percentage of the population cannot afford many of the modern and successful allergy medicines, such as biologics and sublingual immunotherapy, due to their high cost. This can restrict market penetration and patient access to optimal treatment.
Lack of Awareness and Access to Specialty Care: Failures in diagnosis and access to suitable medicines can result from a lack of knowledge about allergy treatment options and the significance of consulting specialists. This is especially important in remote locations where access to specialized medical treatment is limited.
Safety Concerns and Side Effects: Despite their effectiveness, some of the most recent allergy treatments have possible side effects or safety issues that could discourage patients or healthcare professionals from using them. Expanding the market requires addressing these issues through research and teaching.
Notable Recent Updates
- March 2022, The US FDA gave Perrigo Company plc final approval to sell Nasonex 24HR Allergy (mometasone furoate monohydrate 50 mcg) over-the-counter.
- August 2022, the only approved fixed-dose combination medication for seasonal allergic rhinitis was introduced to the US market by Glenmark Pharmaceuticals' arm.
Healthcare Policies and Regulatory Landscape
The national regulatory body in charge of ensuring the efficacy, safety, and calibre of pharmaceuticals and other health products in Canada is Health Canada.Prior to being marketed and sold in Canada, new medications must be reviewed and approved by Health Canada. This also applies to allergy therapy medications, which, in order to guarantee their efficacy and safety, go through a stringent review process.
The Pharmaceutical Drugs Directorate (formerly the Therapeutic Products Directorate) is the division of Health Canada that is especially in charge of drug regulation. The PDD reviews drug applications to evaluate their quality, safety, and efficacy, including allergy therapeutic medication submissions.
Competitive Landscape
Key Players
- Sanofi
- Novartis
- GlaxoSmithKline (GSK)
- AstraZeneca
- Merck & Co.
- Johnson & Johnson
- Roche
- Teva Pharmaceutical Industries
- ALK-Abelló
- Stallergenes Greer.
1. Executive Summary
1.1 Disease Overview
1.2 Global Scenario
1.3 Country Overview
1.4 Healthcare Scenario in Country
1.5 Patient Journey
1.6 Health Insurance Coverage in Country
1.7 Active Pharmaceutical Ingredient (API)
1.8 Recent Developments in the Country
2. Market Size and Forecasting
2.1 Epidemiology of Disease
2.2 Market Size (With Excel & Methodology)
2.3 Market Segmentation (Check all Segments in Segmentation Section)
3. Market Dynamics
3.1 Market Drivers
3.2 Market Restraints
4. Competitive Landscape
4.1 Major Market Share
4.2 Key Company Profile (Check all Companies in the Summary Section)
4.2.1 Company
4.2.1.1 Overview
4.2.1.2 Product Applications and Services
4.2.1.3 Recent Developments
4.2.1.4 Partnerships Ecosystem
4.2.1.5 Financials (Based on Availability)
5. Reimbursement Scenario
5.1 Reimbursement Regulation
5.2 Reimbursement Process for Diagnosis
5.3 Reimbursement Process for Treatment
6. Methodology and Scope
Canada Allergy Therapeutics Market Segmentation
By Treatment Type
- Anti-allergy drugs
- Immunotherapy
By Type of Allergy
- Eye allergy
- Asthma
- Skin allergy
- Food allergies
- Rhinitis
- Other allergy types
By Route of Administration
- Oral
- Inhalers
- Intranasal
- Other routes of administration
By Distribution Channel
- Hospital pharmacies
- Retail pharmacies
- Online pharmacies
- Other distribution channel
Methodology for Database Creation
Our database offers a comprehensive list of healthcare centers, meticulously curated to provide detailed information on a wide range of specialties and services. It includes top-tier hospitals, clinics, and diagnostic facilities across 30 countries and 24 specialties, ensuring users can find the healthcare services they need.
Additionally, we provide a comprehensive list of Key Opinion Leaders (KOLs) based on your requirements. Our curated list captures various crucial aspects of the KOLs, offering more than just general information. Whether you're looking to boost brand awareness, drive engagement, or launch a new product, our extensive list of KOLs ensures you have the right experts by your side. Covering 30 countries and 36 specialties, our database guarantees access to the best KOLs in the healthcare industry, supporting strategic decisions and enhancing your initiatives.
How Do We Get It?
Our database is created and maintained through a combination of secondary and primary research methodologies.
1. Secondary Research
With many years of experience in the healthcare field, we have our own rich proprietary data from various past projects. This historical data serves as the foundation for our database. Our continuous process of gathering data involves:
- Analyzing historical proprietary data collected from multiple projects.
- Regularly updating our existing data sets with new findings and trends.
- Ensuring data consistency and accuracy through rigorous validation processes.
With extensive experience in the field, we have developed a proprietary GenAI-based technology that is uniquely tailored to our organization. This advanced technology enables us to scan a wide array of relevant information sources across the internet. Our data-gathering process includes:
- Searching through academic conferences, published research, citations, and social media platforms
- Collecting and compiling diverse data to build a comprehensive and detailed database
- Continuously updating our database with new information to ensure its relevance and accuracy
2. Primary Research
To complement and validate our secondary data, we engage in primary research through local tie-ups and partnerships. This process involves:
- Collaborating with local healthcare providers, hospitals, and clinics to gather real-time data.
- Conducting surveys, interviews, and field studies to collect fresh data directly from the source.
- Continuously refreshing our database to ensure that the information remains current and reliable.
- Validating secondary data through cross-referencing with primary data to ensure accuracy and relevance.
Combining Secondary and Primary Research
By integrating both secondary and primary research methodologies, we ensure that our database is comprehensive, accurate, and up-to-date. The combined process involves:
- Merging historical data from secondary research with real-time data from primary research.
- Conducting thorough data validation and cleansing to remove inconsistencies and errors.
- Organizing data into a structured format that is easily accessible and usable for various applications.
- Continuously monitoring and updating the database to reflect the latest developments and trends in the healthcare field.
Through this meticulous process, we create a final database tailored to each region and domain within the healthcare industry. This approach ensures that our clients receive reliable and relevant data, empowering them to make informed decisions and drive innovation in their respective fields.
To request a free sample copy of this report, please complete the form below.
We value your inquiry and offer free customization with every report to fulfil your exact research needs.